Research at OFN
Research is an important part of the work of OFN. Our aim is to develop new and improved methods of treatment for movement disorders, neuropathic pain, and any other condition amenable to treatment with implanted nervous system devices.
Participating in research
If you are a patient coming for treatment it is quite likely that we will invite you to take part in one of our research studies. If so, we will provide you with information explaining the study, and you will have the opportunity to ask any questions you wish to about the study before deciding if you would like to take part. You will only be invited to take part in studies that have received approval from the appropriate Research Ethics Committee.
You are under no obligation whatsoever to take part in research. If you do not wish to be involved in any research projects please let us know. You do not need to give any reason for your decision and please rest assured that it will not affect your treatment in any way.
What types of research do we do?
Broadly speaking, we do two types of research that involve patients. Firstly there is "basic science" research and secondly clinical trials.
Basic science
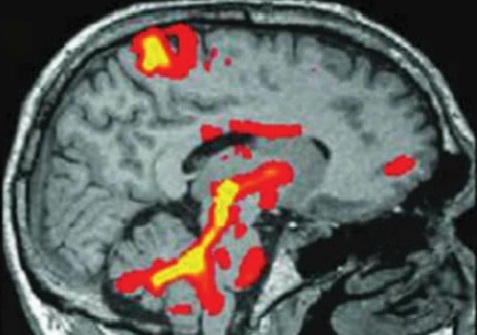
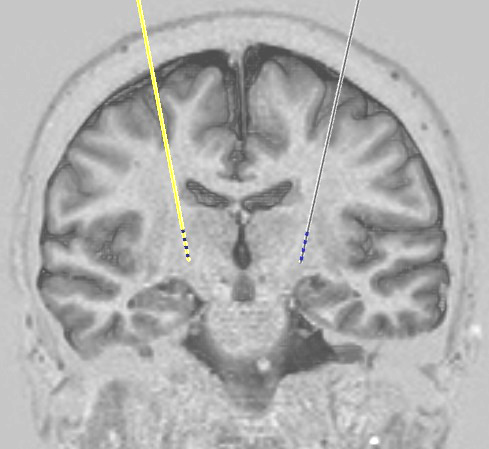
This type of research is mainly aimed at improving our understanding of the conditions that we treat. For example, scientists still do not know all the details of how conditions like Parkinson's disease or dystonia affect the way the brain works, and deep brain stimulation provides a unique opportunity to gain new insights into this because we can use the stimulator electrodes that we have put in to 'listen' to what the brain is doing by recording electrical activity through them. We can also learn things by testing exactly how stimulation changes things like hand movements and eye movements, which we can measure very accurately. These are just examples and the details of what any particular study might involve will be clearly explained if you are invited to take part.
Links to pages describing some of our current areas of research interest can be found on the menu bar at the top of this page.
Clinical trials
Clinical trials are where new or modified treatments are tested on patients. Trials are usually done in order to:
- see how well a new treatment works, or
- compare a newer version of a treatment with an older established version to see whether the newer version is better
There are safeguards built into the design of clinical trials to ensure that patients are not disadvantaged by taking part. If you are invited to take part in a clinical trial we will provide full information about the trial and answer any questions you have about any aspect of the study.
Please see our trials page for details of current and upcoming studies.
Who will benefit from your participation?
Basic science research is unlikely to be of direct benefit to you. The idea is that the knowledge gained will allow us to develop better treatments for future patients with the same condition that you have. In some cases the research may also yield results that might benefit people with other conditions, for example we are studying an effect of deep brain stimulation on blood pressure that we first noticed by chance while looking at the effects of deep brain stimulation for pain.
Clinical trials may benefit their participants, if the intervention being tested represents a significant improvement over the previous standard treatment.
Publications
A list of research papers, review articles, books and book chapters authored by members of OFN.
Copyright OFN Wednesday, 11 February, 2015.